|
Back to Blog
Capto plasmid select7/25/2023  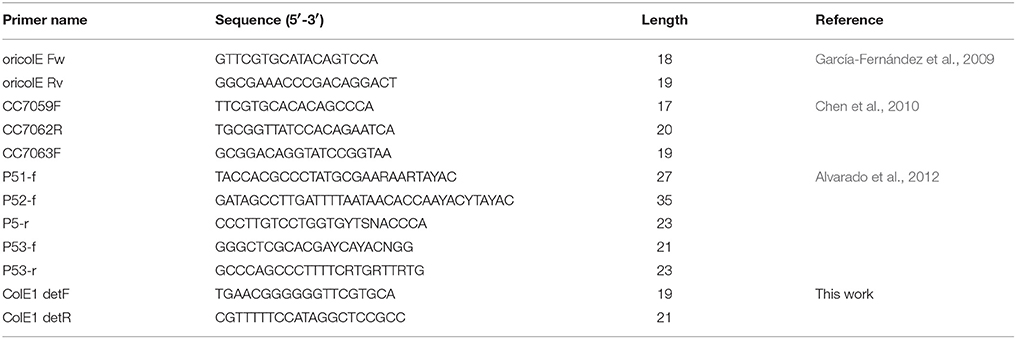
Barstar does not play any role in barnase expression. 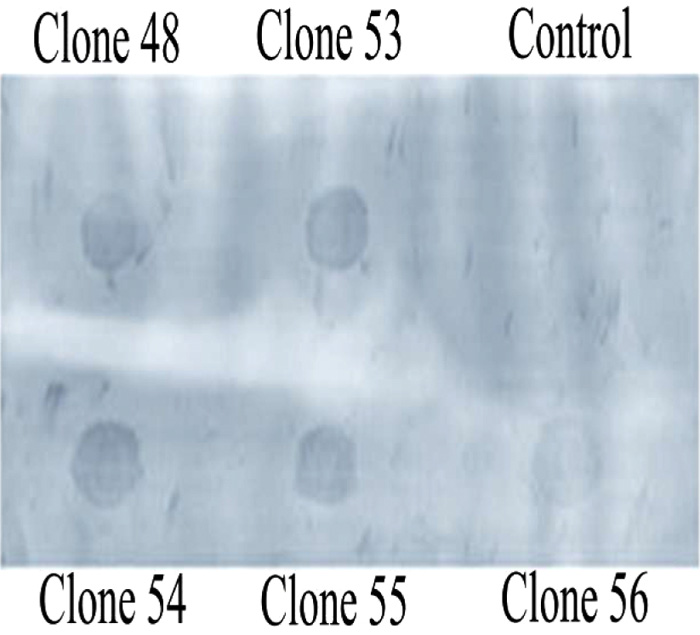
Due to its importance in inactivating barnase in the cell, barstar is cytoplasmic in the Bacillus sp. It binds to the active site and neutralizes any RNase that remains behind in the cytoplasm and thereby averts the danger to host cell-RNA. Barstar (89 residues), a small 10 kDa cytoplasmic protein, was the first discovered inhibitor protein specific for guanyl-specific RNases such as barnase and binase I. amyloliquefaciens, an intracellular inhibitor of barnase called barstar is produced which tightly binds to barnase leading to inhibition and thereby protection of the host cell from the toxic effects of the RNase. This emphasizes the significance of the corresponding intracellular inhibitor for successful recombinant expression as well. However, expression of this gene in the cytoplasm is toxic to the host cell when a corresponding inhibitor is not co-expressed. The simple structure of the enzyme also allows its active expression in a wide variety of cell types. This led to the idea of expressing the protein recombinantly in its active conformation in the cytoplasm just as well as in the periplasmic space of a host cell such as Escherichia coli. The activity was reported in the pH range of 7.5 to 9.3 with a maximum at 8.5. Barnase catalyses the cleavage of single-stranded RNA through hydrolysis of the phosphodiester bonds at GpN sites, with a higher rate for GpG and GpA sites. It contains no disulfide bonds, divalent cations as cofactors or other non-peptide moieties. 3.1.27) produced and secreted by Bacillus amyloliquefaciens, is a small single-chain RNase of 110 amino acids with a molecular mass of 12.3 kDa. Application of even a relatively small amount of recombinant RNase would contribute to greatly reducing the initial RNA levels in alkaline lysates thereby augmenting further downstream plasmid purification steps.īarnase (EC No. Also important is to curtail the massive enzyme loss during purification by cation exchange chromatography. The current work focusses on the downstream purification strategies for a recombinant RNase and sets a framework for higher scale production if specific productivity is increased by optimal hosts and/or re-engineered plasmids. Currently, this is capable of processing small amounts (13 g) of bacterial biomass for plasmid production. Although current levels of volumetric activity achieved are quite meagre (4 Kunitz units mL − 1), in principle its application to plasmid DNA purification could be proved. This was followed by hydrophobic interaction chromatography which resulted in a concentrated fraction of active enzyme. 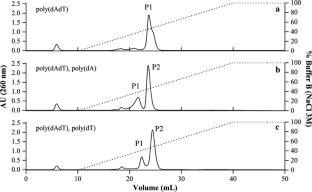
Cation exchange chromatography was employed as a primary purification step. The pure enzyme without the inhibitor barstar was exported to the extracellular space through the periplasm and then purified from the cell-free supernatant. The current study describes a plasmid construct that allowed expression of barnase in Escherichia coli under co-expression of its native inhibitor barstar. The recombinant expression of bacterial RNase is fraught with toxicity problems making it a challenging enzyme to express. As the field of gene therapy now enters the clinical stage, such enzymes need to be phased out or alternative purification protocols need to be developed to ensure product safety and regulatory compliance. The use of bovine-origin ribonucleases has been part of the standard protocol for plasmid DNA purification. 
0 Comments
Read More
Leave a Reply. |
 RSS Feed
RSS Feed
